
Incomplete biomarker testing can lead to inappropriate treatment selection
Fewer than 20% of advanced NSCLC patients get complete guideline-recommended biomarker testing9
10
The number of biomarkers guidelines recommend testing advanced NSCLC patients for before treatment
The list of biomarkers that are matched with an FDA-approved targeted therapy is growing.
EGFR
KRAS
ALK
ROS1
BRAF
MET
RET
ERBB2
NTRK
PD-L1
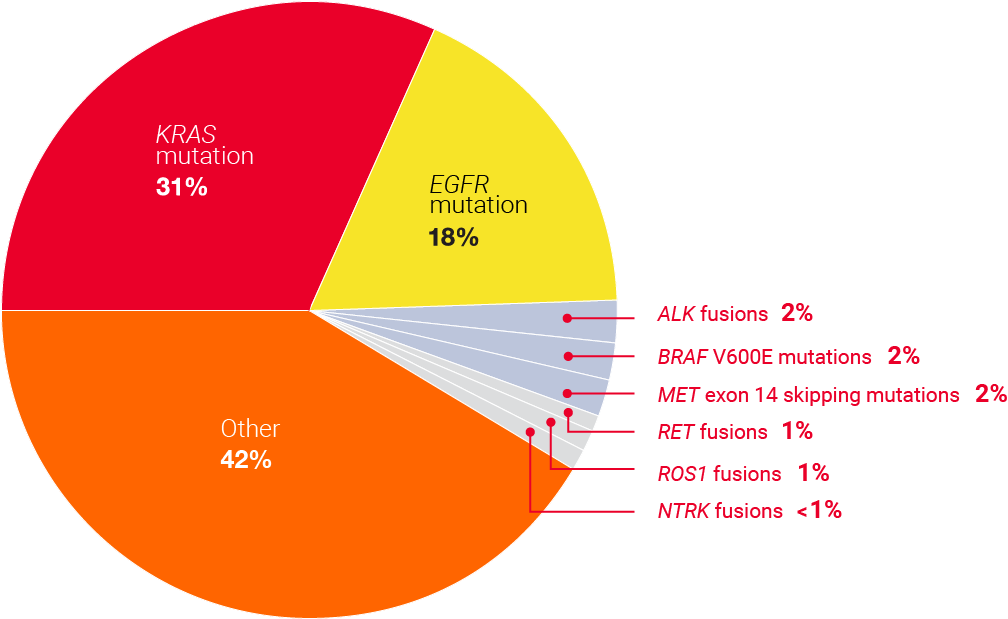
2 in 3 NSCLC patients
have a biomarker that can be targeted with available therapies10
Matching advanced NSCLC patients to targeted therapy using biomarker testing results improves outcomes11
Sotorasib (LUMAKRAS™)18 significantly
increases PFS compared to docetaxel11
Sotorasib targets KRAS G12C, one of the most common NSCLC biomarkers. 1 in 8 patients with advanced lung cancer has a KRAS G12C mutation.10
Advanced NSCLC patients with targetable mutations should not be treated with immunotherapy1
82%
of NSCLC patients could get the wrong treatment because their doctors fail to order complete biomarker testing.9
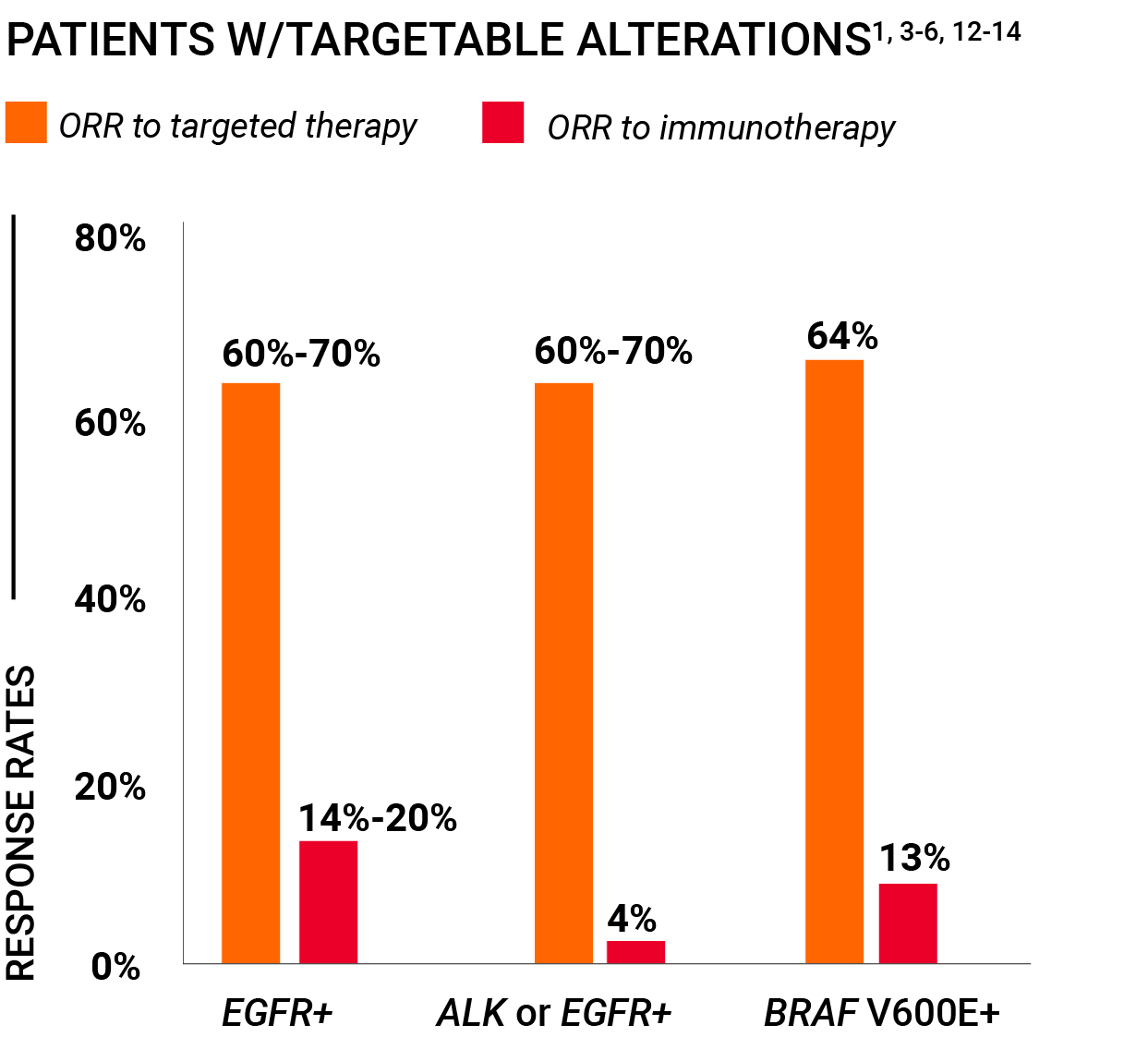
One chance to make the right decision
There may be just one opportunity for a doctor to make the right decision about a patient’s first-line treatment for advanced NSCLC.